|
1/13/2024 0 Comments Leap therapeutics 
Since 2020, the clinical-stage biotechnology company has been developing multiple therapeutic platforms to enhance T cell (Treg) function, including biologics and cell therapies.ĬOYA 301 is an immunomodulatory cytokine for subcutaneous administration intended to enhance regulatory Treg function in vivo to treat the systemic and neuro-inflammation underlying certain autoimmune and neurodegenerative diseases. for the development and commercialization of COYA 301, Coya’s low-dose IL-2 subcutaneous administration product candidate. 
To help make a difference, players like Coya are actively ramping up and expanding their therapies to address the needs of the market.Ĭoya just announced the expansion of their exclusive worldwide rights with ARScience Biotherapeutics Inc. The statistics around autoimmune diseases are certainly worrying. Expansion Of Its Exclusive Worldwide Rights Autoimmune diseases are among the top 10 leading causes of death among American women. 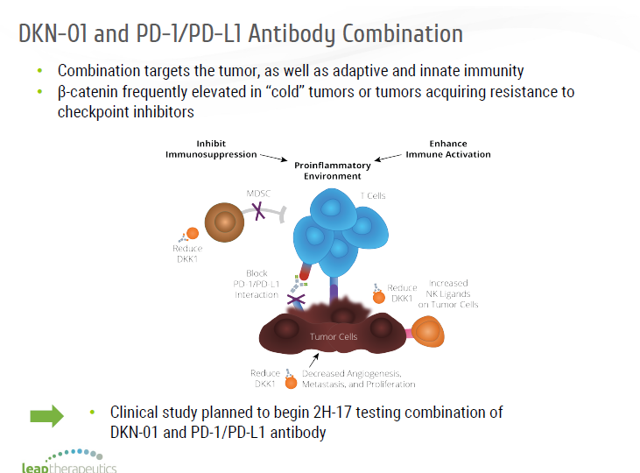
More worryingly, AARDA further said approximately 75% of those affected are women and tend to cluster in families, impacting multiple family members and generations. A deep dive into the statistics by the American Autoimmune Related Diseases Association (AARDA) shows that an estimated ~50 million Americans have one or more autoimmune diseases. COYA has just taken a giant leap in making effective proprietary treatments focused on certain neurodegenerative and autoimmune diseases available to more patients, as it announced the expansion of its exclusive worldwide rights for the development and commercialization of COYA 301 - the company’s low-dose IL-2 subcutaneous administration product candidate.įor industry watchers, including investors, this might be very important, especially as multiple autoimmune diseases have largely unmet medical needs. Analysts typically rate each stock once per quarter.Houston, Texas-based Coya Therapeutics Inc. Analysts may attend company conference calls and meetings, research company financial statements, and communicate with insiders to publish "analyst ratings" for stocks. About Analyst RatingsĪnalysts work in banking and financial systems and typically specialize in reporting for stocks or defined sectors. Its other clinical studies include P100, P102, P204,and P205. The company is studying DKN-01 in multiple ongoing clinical trials in patients with esophagogastric cancer, hepatobiliary cancer, gynecologic cancers, or prostate cancer. DKK1 is a protein that regulates the Wnt signaling pathways and enables tumor cells to proliferate and spread, as well as suppresses the immune system from attacking the tumor. Its clinical stage program is DKN-01, a monoclonal antibody that inhibits Dickkopf-related protein 1, or DKK1. Leap Therapeutics Inc is a biopharmaceutical company developing novel therapies designed to treat patients with cancer by inhibiting fundamental tumor-promoting pathways and by harnessing the immune system to attack cancer cells. Shares of Leap Therapeutics are trading down 1.21% over the last 24 hours, at $0.49 per share.Ī move to $2.50 would account for a 408.85% increase from the current share price. has decided to maintain its Buy rating of Leap Therapeutics (NASDAQ:LPTX) and lower its price target from $4.00 to $2.50.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
